The world
needs
better
vaccines.
needs
better
vaccines.
We’re
striving
to create
them.
striving
to create
them.
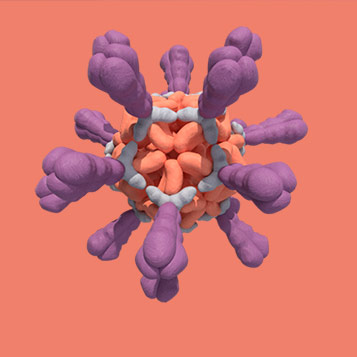
A novel approach centered around virus-like particles (VLPs)
We’re a clinical-stage company working to develop a new kind of vaccine that can protect vulnerable older adults from multiple respiratory viruses. Underpinning this work is a unique approach that utilizes virus-like particles (VLPs). Our VLPs are intentionally designed to mimic the structure of viruses in order to empower a better immune response. They may offer potential advantages vs. currently available modalities, including magnitude of response, breadth of coverage including related viral strains and mutations, longer durability, lower incidence of side effects, easier combinability to allow multiple targets in one vaccine, and more.